U.S. to widen COVID antiviral pill distribution
- 26-Apr-2022
The administration expects to increase its direct distribution to over 30,000 locations soon and reach 40,000 sites over the coming weeks.
WHO recommends Merck’s COVID pill for high-risk patients
- 03-Mar-2022
A World Health Organization (WHO) panel on Wednesday backed the use of molnupiravir- Merck & Co’s COVID-19 antiviral pill- for high-risk patients. The expert panel conditionally recommended the pill, molnupiravir, for patients with non-severe disease who are at high risk of hospitalisation, such as the immunocompromised, the unvaccinated, older people and those with chronic diseases. […]
China approves use of Pfizer’s COVID drug Paxlovid
- 12-Feb-2022
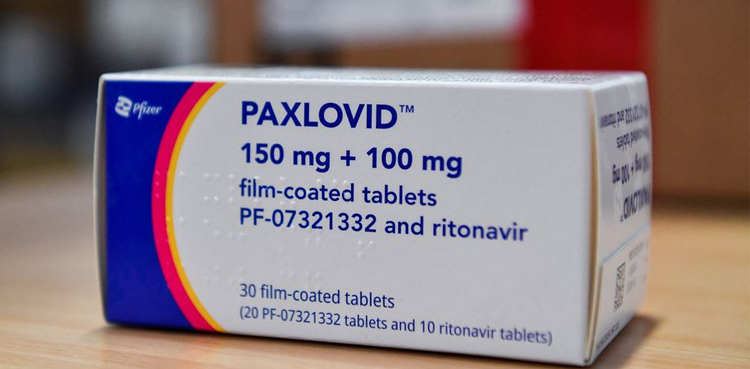
China’s medical products regulator said on Saturday it has given conditional approval for Pfizer’s COVID-19 drug Paxlovid, making it the first oral pill specifically developed to treat the disease cleared in the country. The National Medical Products Administration said Paxlovid is approved to treat adults who have mild to moderate COVID-19 and high risk of […]
Shionogi COVID pill about to get approval in Japan
- 07-Feb-2022
Japanese Prime Minister Fumio Kishida said on Monday the government would consider granting conditional early approval for the oral COVID-19 treatment being developed by Shionogi & Co, as the firm prepares to start a late-stage global trial. Shionogi Chief Executive Isao Teshirogi told reporters that the company could file in Japan for early approval of […]
UK to begin testing Merck’s COVID pill for hospitalised patients
- 24-Jan-2022
UK scientists will begin testing Merck and Ridgeback Biotherapeutics’ antiviral pill molnupiravir as a possible treatment for patients hospitalised with COVID-19, amid the worldwide spread of the Omicron variant. The pill is approved in Britain for use in people with mild to moderate COVID-19, but it is not known whether it would work in patients […]
Cheap version of Merck COVID pill to be made for poorer nations
- 20-Jan-2022
A UN-backed agency has struck a deal for nearly 30 generic drugmakers to make low-cost versions of Merck’s COVID-19 pill molnupiravir for poorer nations, widening access to a drug seen as a weapon in fighting the pandemic. The antiviral drug, which in December received emergency approval in the United States, reduces hospitalizations and deaths of […]
US regulator authorizes Merck’s Covid pill
- 23-Dec-2021
The US Food and Drug Administration (FDA) on Thursday authorized Merck’s Covid pill for high risk adults, a day after giving the green light to a similar pill by Pfizer. “Today’s authorization provides an additional treatment option against the COVID-19 virus in the form of a pill that can be taken orally,” said FDA scientist […]
Pfizer says Covid pill drastically reduces severe disease
- 14-Dec-2021
Pfizer said Tuesday that clinical trials confirmed its Covid pill, a new type of antiviral treatment that should withstand the mutations seen with Omicron, drastically reduced hospitalizations and deaths among at-risk people by almost 90 percent. The announcement came as a real-world study from South Africa showed two doses of the Pfizer-BioNTech vaccine was 70 […]