Trump links autism to Tylenol and vaccines, claims not backed by science
- 23-Sep-2025
U.S. President Donald Trump on Monday linked autism to childhood vaccine use and the taking of popular pain medication Tylenol by women when pregnant, elevating claims not backed by scientific evidence to the forefront of U.S. health policy. In an extraordinary news conference at the White House, the Republican president delivered medical advice to pregnant […]
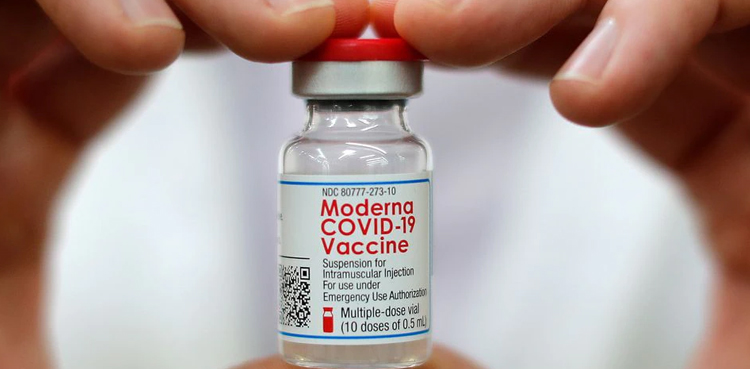
Moderna files for review of updated COVID vaccine
- 24-May-2025
Moderna said on Friday it has filed a marketing application for the review of its updated COVID-19 vaccine with the U.S. Food and Drug Administration. The company said the submission for the vaccine, branded as Spikevax, is based on guidance from the FDA, which advised that the shots should be updated to target strains that […]
Hajj 2025: MoRA to start vaccine provision to intending pilgrims from today
- 21-Apr-2025
The Ministry of Religious Affairs will start the provision of vaccine to intending Hajj 2025 pilgrims from Monday (today). The vaccine will be provided in Islamabad, Lahore, Peshawar and Multan from Monday. In Karachi, Rahim Yar Khan, Sukkur, Faisalabad and Sialkot, Hajj pilgrims will receive vaccine from Tuesday. In Quetta, mandatory vaccination to Hajj pilgrims […]
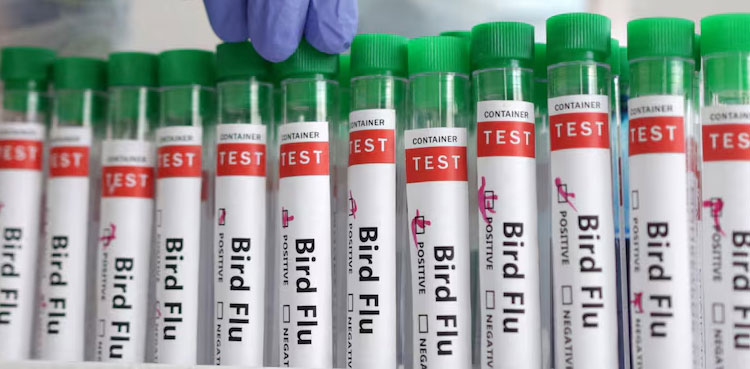
Zoetis’ bird flu vaccine gets conditional approval in US
- 15-Feb-2025
The U.S. Department of Agriculture has given a conditional approval to Zoetis to use its bird flu vaccine in poultry, the animal healthcare company said on Friday. Bird flu has infected nearly 70 people in the United States, with one death, since last April. Most of those infections have been among farm workers exposed to […]
Saudi Arabia suspends meningitis vaccine requirement for travellers
- 07-Feb-2025
KARACHI: Pakistani travellers heading to Saudi Arabia for Umrah or Hajj are no longer required to provide proof of Neisseria meningitis vaccination, as the Saudi Civil Aviation Authority has suspended the requirement, ARY News reported. Saudi General Authority of Civil Aviation (GACA) issued a new circular on Thursday for travellers from various countries. According to […]
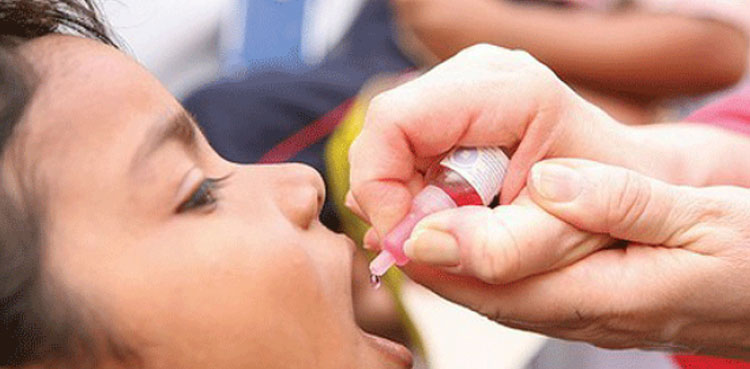
‘Over 1.1mn children left unvaccinated in Pakistan’s 2024 anti-polio drive’
- 11-Jan-2025
ISLAMABAD: The final sub-national polio campaign in Pakistan for the year 2024 has not reached its intended target and a total of 1,122,537 children remained unvaccinated, ARY News reported citing sources. The campaign primarily aimed to vaccinate 36,360,037 children, but only 35,712,922 children were successfully vaccinated which indicates that 98 per cent of the vaccination […]
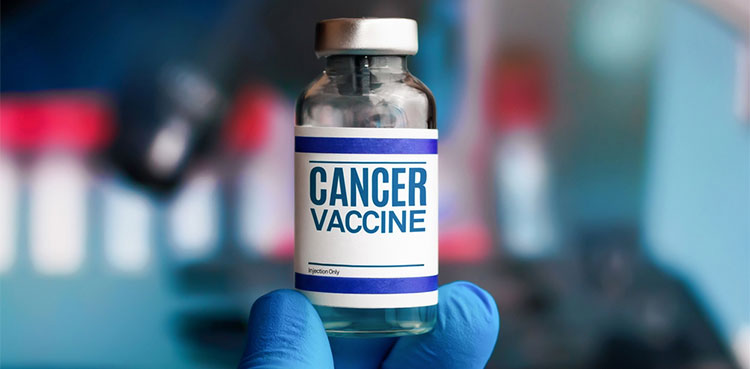
Russia claims to develop own cancer vaccine
- 18-Dec-2024
Russia’s Ministry of Health has announced the development of a cancer vaccine that will be provided to patients at no cost. As reported by the state media, Andrey Kaprin, the director of the Radiology Medical Research Center within the Ministry, indicated that the vaccine is set to be introduced in early 2025. This vaccine is […]
US expands bird flu testing after finding symptom-free infections in people
- 09-Nov-2024
Farm workers who have been exposed to animals with bird flu should be tested for the virus even if they do not have symptoms, the U.S. Centers for Disease Control (CDC) and Prevention said on Thursday. The change to the agency’s testing recommendation comes as the U.S. Department of Agriculture is also expanding its testing […]
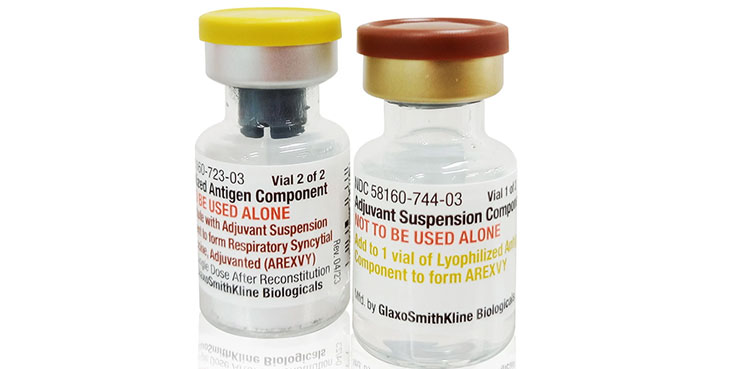
Arexvy: Canada approves GSK’s RSV vaccine
- 05-Nov-2024
GSK has received Canadian approval for Arexvy, its respiratory syncytial virus vaccine in adults between 50 and 59, the company said on Tuesday, making it the country’s first authorized RSV shot for that age group. The U.S. Centers for Disease Control and Prevention decided to hold off recommending the use of the vaccine, Arexvy, in […]
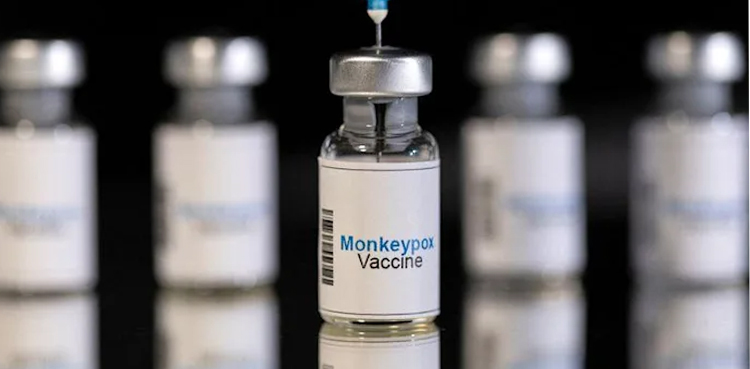
Congo requests mpox vaccines from Japan
- 19-Aug-2024
Japan’s Ministry of Health, Labour and Welfare said on Monday it has received a request from the Democratic Republic of the Congo for the provision of mpox vaccines and needles manufactured in the country. This comes days after the World Health Organization declared mpox a global public health emergency for the second time in two […]